 Goemkarponn desk
Goemkarponn desk
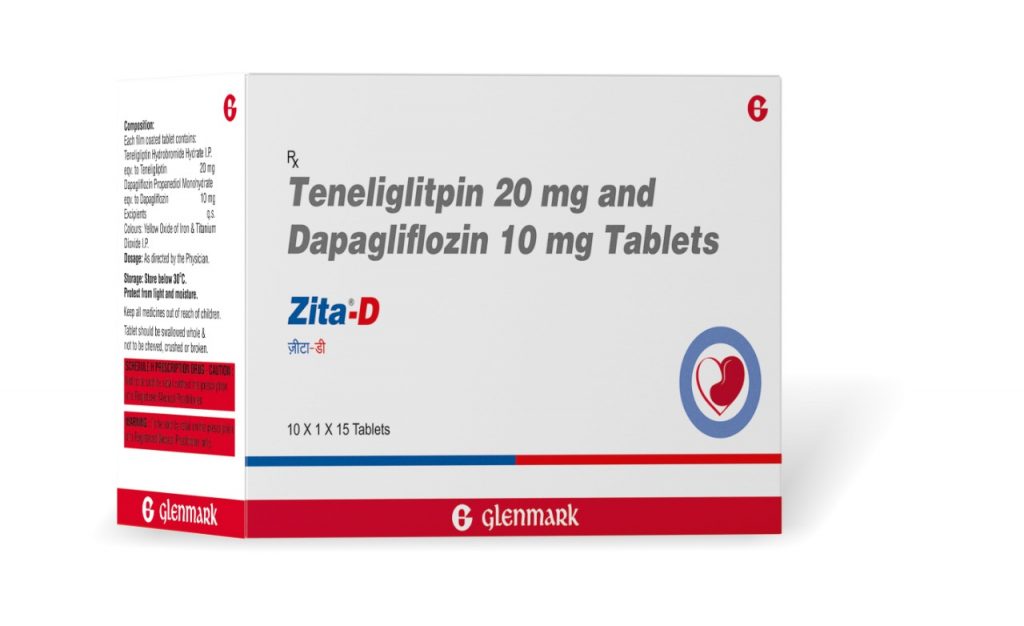
PANAJI: Glenmark Pharmaceuticals Limited (Glenmark), an innovation-driven global pharmaceutical company, became the first to launch Teneligliptin (20mg) + Dapagliflozin (5mg/10mg) fixed-dose combination (FDC) for the treatment of adult patients with type 2 diabetes, especially the ones with comorbidities. Marketed under the brand name Zita D; it contains Teneligliptin (20mg) + Dapagliflozin (5 mg/10 mg), and must be taken once daily under prescription to improve glycemic control & prevent complications in adult patients with type 2 diabetes, especially the ones with comorbidities.
On the launch, Alok Malik, EVP & Business Head – India Formulations, Glenmark Pharmaceuticals, said, “Diabetes is growing in India at an alarming rate, and eight out of every ten diabetic patients suffer from comorbidities. Glenmark being a leader in the diabetes segment in India is proud to bring Zita D, a well-researched and affordable fixed dose combination, which will significantly improve the glycemic control in adult patients suffering from uncontrolled Type 2 diabetes, especially the ones with comorbidities.”
Teneligliptin is a widely used DPP4i (Dipeptidyl Peptidase 4) inhibitor, whereas Dapagliflozin is the most prescribed SGLT-2i (Sodium-glucose co-transporter 2) inhibitor for diabetic patients in the country. While Zita D is a promising treatment option in managing uncontrolled Type 2 diabetes with comorbidities, this FDC has also proven to be effective among adult diabetic patients without comorbidities. Glenmark’s Zita D has priced at around INR 14 per tablet for FDC of Teneligliptin 20mg + Dapagliflozin 5mg, and INR 15 per tablet for FDC of Teneligliptin 20 mg + Dapagliflozin 10 mg, per day.
Glenmark has a strong legacy of bringing new and effective treatment options for diabetic patients. In 2015, Glenmark revolutionized diabetes treatment in India by being the first to launch the DPP4 inhibitor – Teneligliptin, followed by a fixed-dose combination of Teneligliptin + Metformin. In continuation towards its legacy, Glenmark launched Remogliflozin, a novel SGLT-2 inhibitor, in 2019 after its first global approval in India and subsequently launched its combinations with metformin and Vildagliptin. Earlier in 2022, Glenmark launched Sitagliptin and its FDCs, followed by Lobeglitazone.
Sorry, there was a YouTube error.